TM 10-6140-200-14
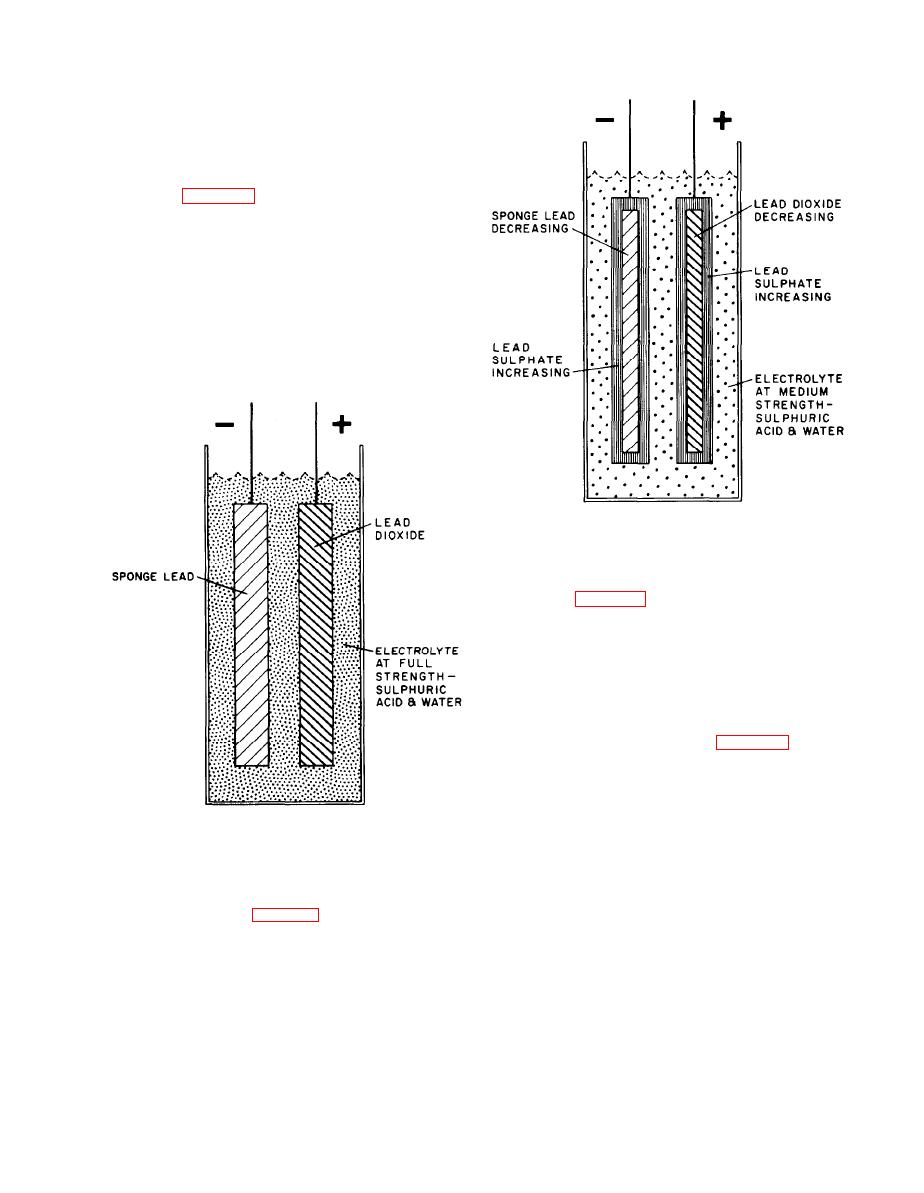
a. The Fully Charged Cell or Battery. The positive and
negative plates, or electrodes, are separated from each
other and immersed in electrolyte. In the fully charged
condition the active material of the positive plate is lead
dioxide and that of the negative plate is sponge lead as
indicated in Figure 1-6. The electrolyte is a solution of
sulfuric acid and water that normally varies in specific
gravity from 1.275 to 1.295. The combination produces a
voltage of approximately 2 volts on open circuit. This
voltage potential results from the fundamental charac-
teristic of a storage battery which dictates that when two
electrodes of dissimilar metals are immersed in suitable
electrolyte, and a circuit is closed between the two,
electrons begin to flow. A fully charged cell should
normally have an on-charge voltage of from 2.45 to 2.70
volts when charging at the finish rate.
Figure 1-7. Discharging cell
water. Almost all of the active material of both positive
and negative plates is converted to lead sulfate as
illustrated by Figure 1-8, and an effective electrochemical
reaction is no longer possible. At this point the battery
has reached its discharge limit.
d. The Charging Cell or Battery. The charging action
begins when the terminals of the battery are connected to
an external source of direct current. The electrochemical
reaction is reversed and the positive plates, negative
plates, and electrolyte start returning to their original
charged condition as indicated by Figure 1-9. Charging
causes the battery voltage to rise as active materials are
restored. A cell being charged may have a voltage of from
2.12 to 2.70 volts depending upon charging rate and time.
e. General. From the above it can be seen that storage
Figure 1-6. Fully charged cell
batteries do not actually store electrical energy. Instead,
they accept the electrical energy delivered to them during
charging and convert it into chemical energy. During
being discharged or used, lead dioxide and sponge lead
discharging, this chemical energy is reconverted into
combine with sulfuric acid to form lead sulfate within
electrical energy to be used as needed.
both plates as shown in Figure 1-7. This action causes the
specific gravity of the electrolyte to decrease. As the
f. As an operating guide, to obtain the best per-
discharge progresses, individual cell and battery voltage
formance and life from a motive power storage battery,
declines, generally in direct proportion to the rate of
the depth of discharge should not regularly exceed 80%
discharge.
of the battery's rated capacity in ampere-hours. It should
be charged after each shift of use or whenever the specific
gravity of the electrolyte falls below 1.240. It is very
c. The Discharged Cell or Battery. As the depth of
important that proper ventilation be provided during
discharge increases more sulphuric acid is removed from
charging to make certain that (1) the hydrogen gas, given
the electrolyte so the specific gravity decreases and may
off toward the end of the charging process, is dissipated
drop below 1.100 as it approaches the specific gravity of
1-5



