TM11-6140-203-14-1
2-3. Electrochemical Action
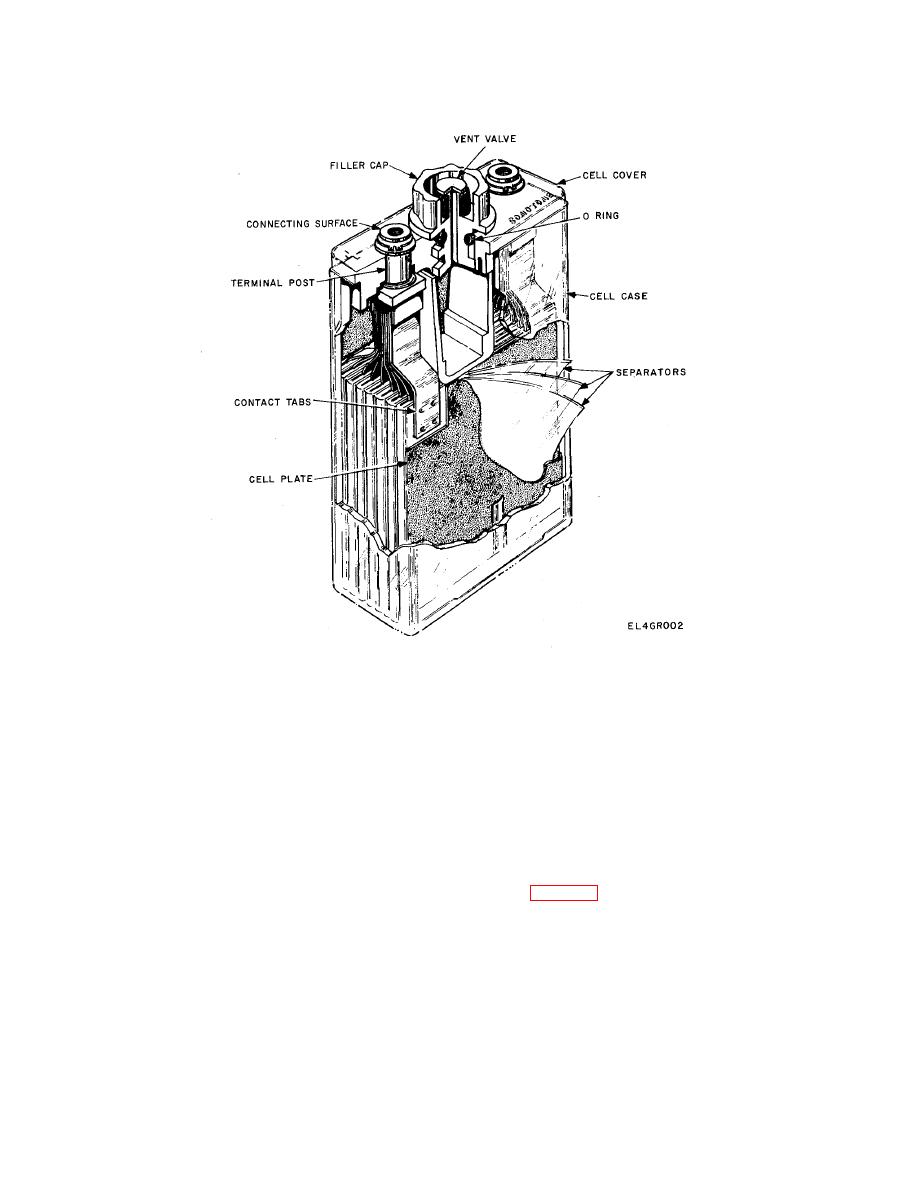
(5) Cell vent. Each cell is equipped with a vent
plug and filler cap. These vents may be either a one-
The exact chemical reactions that occur within a cell of
piece or a two-piece assembly. The cell vent can be re-
the nickel-cadmium battery during charge and dis-
moved for cleaning and adjustment of electrolyte
charge are open to question, particularly with regard
level. When excessive gases develop in a cell during
to the reduced and oxidized states of the active
charge, they escape through the vent hole. The vent re-
materials. However, the essential operation can be de-
lieves gas pressure by remaining closed until a pres-
scribed. Figure 2-2 illustrates the essential electro-
sure of at least 2 psi is reached and will open at a pres-
chemical actions.
sure below 10 psi. Except when releasing gas, the vent
a. Charge. When charging current is applied to the
remains sealed to prevent electrolyte leakage, the
cell, the cadmium-oxide material of the negative plates
entry of foreign material into the cell or contamina-
gradually loses oxygen and becomes metallic cadmium
tion of the electrolyte by exposure of air high in carbon
and the nickel-oxide active material of the positive
dioxide content. When a two-piece cell vent is used,
plates is brought to a higher state of oxidation. These
the vent valve may be removed for shipping and re-
changes continue in both sets of plates as long as the
placed by a shipping screw. Before putting the battery
charging current is applied, or until the active
into service the slipping screw must be removed and
materiels at the plates have been completely con-
replaced by the vent valve.
verted. The cell emits gas toward the end of this proc-



